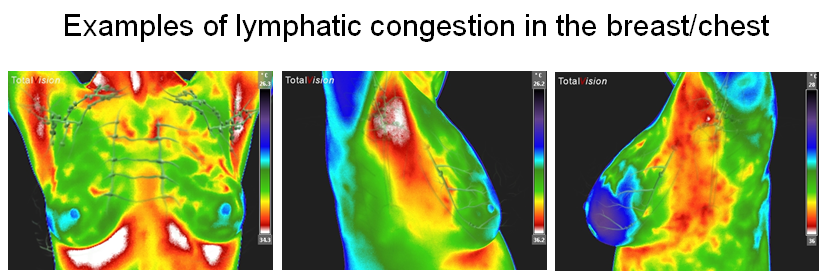
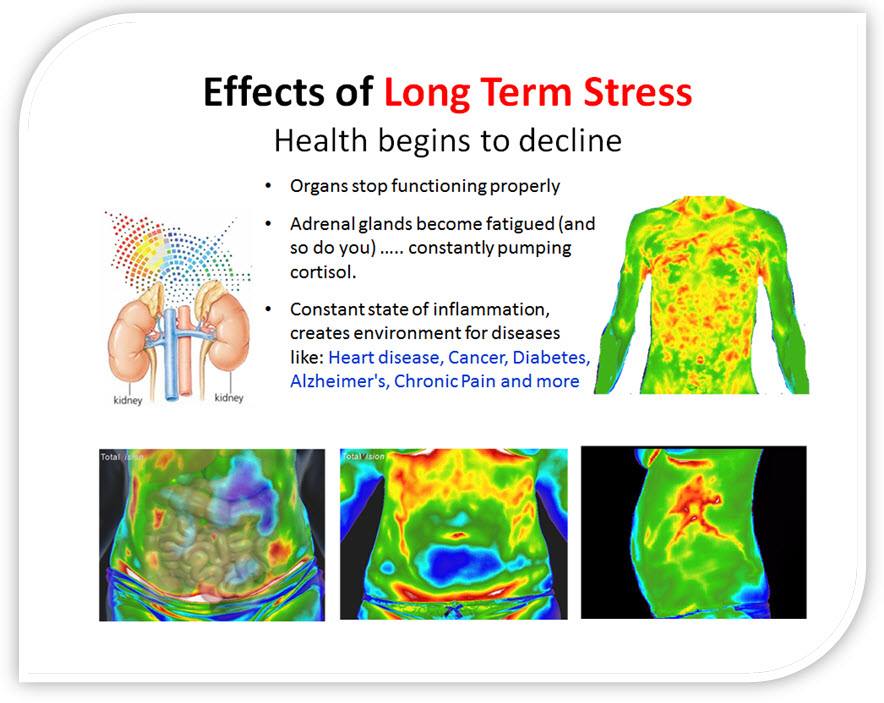
Because something is still missing
According to a BMJ Quality & Safety study, approximately 12 million American adults seeking outpatient care experience misdiagnosis annually — representing 1 in 20 patients. Researchers indicate that roughly half of these cases carry potential for serious harm.
Despite the prevalence of diagnostic errors, thermography remains absent from mainstream medical practice. This absence reflects not a lack of availability, but rather the medical field's reluctance to adopt complementary tools. It's time to embrace something affordable, harmless and different to supplement existing diagnostic methods.
FDA Indication
Thermographic devices are FDA-approved and intended to measure skin temperature patterns across various body regions including the head, neck, breast, chest, abdomen, back, and extremities as an adjunctive diagnostic screening tool.
A Health Discovery Tool
Rather than functioning as a "screening device" targeting specific diseases, thermography operates as a Health Discovery Tool. It offers a non-invasive approach to reveal thermal changes imperceptible through conventional examination.
For healthy individuals: establishing baseline health status and preservation strategies.
For those with existing conditions: uncovering potentially valuable new diagnostic information.
Instead of asking "Why Thermography?", consider "Why Not Thermography?" and schedule a baseline health assessment.

By Carol Chandler